Which compound will migrate the fastest during thin layer chromatography using silica plates?
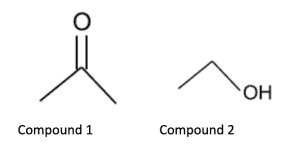
A. Compound 1 because it is more polar and will interact more strongly with the stationary phase
B. Compound 1 because it is more nonpolar and will interact more strongly with the mobile phase
C. Compound 2 because it is more polar and will interact more strongly with the stationary phase
D. Compound 2 because it is more nonpolar and will interact more strongly with the mobile phase
Click for Explanation
Answer choice B is correct. Thin layer chromatography uses a polar silica stationary phase which more strongly interacts with polar molecules. Typically, the mobile phase will be some type of nonpolar solvent. The compound that migrates the fastest will be the one that interacts more strongly with the mobile phase, essentially the compound that is less polar. Compound 1 does have some polarity with the carbonyl, but compound 2 has a hydroxyl group which is more polar. Compound 2 will interact with the stationary phase via hydrogen bonds, slowing its migration. Note that we could eliminate answer choices A and D since they incorrectly assign polarity to the two compounds: 1 is more nonpolar, 2 is more polar.
Want more MCAT practice?
We’ve got options for every schedule and learning style!
Whether you need the flexibility of a Self-Paced Course, the instruction of a live 515+ Course, or the 1:1 attention of a private MCAT tutor, Blueprint MCAT has the MCAT prep option that works for your learning style!
Get started with a free MCAT diagnostic, one free practice exam, and tons more MCAT prep resources.
Not sure which option is right for you? Schedule a free MCAT consultation with an MCAT expert using the form below. No obligation, just expert advice.


