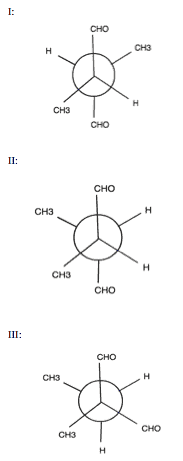
Plane-polarized light will NOT be rotated by which of the following molecules:
A. I only
B. III only
C. II and III only
D. I and III only
Click for Explanation
A. I only
B. III only
C. II and III only
D. I and III only
Plane-polarized light is not rotated if a molecule is meso (like I) or if it has no chiral centers (like III). To see how structure I is meso, remember that in a Newman projection, groups can be rotated freely, since free rotation is possible around a single bond. If the front carbon is spun 180º, each of the substituents lines up with the same group on the rear carbon. Thus, the molecule has a plane of symmetry and is meso.
For structure III, note that the front central carbon is bound to two CHO groups, while the rear central carbon is attached to two hydrogen atoms. Thus, no carbon in the compound is bound to four different substituents, meaning that this compound has no chiral centers. As a result, we know that structures I and III will not rotate plane-polarized light, and D is our answer.
Want more MCAT practice?
We’ve got options for every schedule and learning style!
From the best online MCAT course created by top instructors with 524+ MCAT scores to the most representative full-length practice exams and private tutoring, we can custom tailor your MCAT prep to your goals!
Not sure which option is right for you? Schedule a free MCAT consultation with an MCAT expert using the form below. No obligation, just expert advice.