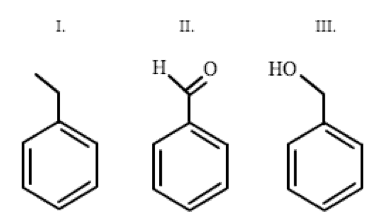
Which of the following correctly orders the boiling point of benzaldehyde, benzyl alcohol, and ethyl benzene from highest to lowest?
- II > III > I
- III > II > I
- I > II > III
- III > I > II
Click for Explanation
This question requires the examinee to compare the boiling point of three compounds based on molecular weight and intermolecular forces. The greater the molecular weight, the higher the boiling point. Also, the stronger the intermolecular forces, the higher the boiling point.
Ethyl benzene forms London dispersion forces and has a molecular weight of 106.2 g/mol. Benzaldehyde has a molecular weight of 106.1 g/mol and forms dipole-dipole forces in addition to London dispersion forces. Lastly, benzyl alcohol has a molecular weight of 108.1 g/mol and forms hydrogen bonds, a special type of dipole-dipole interaction, as well as London dispersion forces.
Since all of the molecular weights are approximately the same, the intermolecular forces of each compound will determine the boiling point. Hydrogen bonds (dipole-dipole) in benzyl alcohol are stronger than dipole-dipole forces in benzaldehyde, and all dipole-dipole forces are stronger than London dispersion forces. Therefore, benzyl alcohol (BP: 205.3oC) > benzaldehyde (BP: 178.1oC) > ethyl benzene (BP: 136.0oC), or III > II > I. Based on these comparisons, B is the correct answer.
Want more MCAT practice?
We’ve got options for every schedule and learning style!
From the best online MCAT course created by top instructors with 524+ MCAT scores to the most representative full-length practice exams and private tutoring, we can custom tailor your MCAT prep to your goals!
Not sure which option is right for you? Schedule a free MCAT consultation with an MCAT expert using the form below. No obligation, just expert advice.

MCAT is a registered trademark of the Association of American Medical Colleges (AAMC), which is not affiliated with Blueprint.