MCAT Orgo Practice Question
Despite being a common source of MCAT anxiety, organic chemistry is actually the least important part of the test. It only makes up about a quarter of the biological sciences section. That means on the 3-45 scale for your overall score, organic chem only accounts for 1/12 of your score. Chemistry and physics are each twice as important, biology is three times as important, and verbal is four times more important than organic chem.
In fact, the first time I took the MCAT (back in the dark ages when it was on paper and you had to ride a dinosaur to the testing location), I had just failed organic chemistry (let’s just say Sophomore year was rough and leave it at that). Despite being a terrible organic chem student, I still managed a 12 on the Biological Sciences section – because biology is so much more important!
Having said all of that, you can’t entirely ignore organic chem. In general, you’re going to get the best bang-for-your-buck by mastering a few commonly tested areas, rather than wasting time trying to memorize tons and tons of less-commonly tested stuff.
Out of the commonly tested areas, nothing beats the most fundamental skill of all: nomenclature. Very often, the test will ask questions that aren’t terribly complex, but can completely frustrate you if you’re not familiar with the nomenclature of functional groups or common molecules. This question is good example of that:
Item 125
A chromatograph is set up in which the stationary phase of the system is one that is susceptible to hydrogen bonding and the mobile phase is non-polar. Which of the following types of compounds would likely move the least in the course of the chromatography?
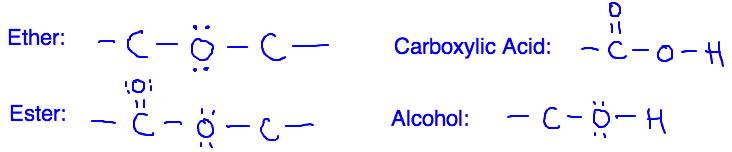
A) Ethers
B) Esters
C) Alcohols
D) Carboxylic Acids
Explanation
This question expects you to be familiar with the concept of intermolecular forces and with the nomenclature of basic functional groups. We see in the question stem that the stationary phase permits hydrogen bonding. We’re then told to find the compound that would move the least. That means picking out the compound that’s going to have the most H-bonding.
If a compound can engage in H-bonding, then it will “stick” to the stationary phase, instead of moving along with the mobile phase. Non-polar (or less-polar) compounds will tend to prefer the mobile phase, so they’ll move farther.
The first thing we can do is eliminate (A) and (B) since neither esters nor ethers can do H-bonding. Those functional groups involve oxygen bonded to carbon, but to do H-bonding we need hydrogen bonded to either nitrogen, oxygen, or flourine.
That leaves us with (C) and (D). Both of these functional groups have a hydrogen bonded to an oxygen, so they can both do H-bonding with the stationary phase in the chromatography. To decide between those choices, we need to remember that a carboxylic acid has two oxygens in it. Those two oxygens are going to create a more polar O-H bond than the one present in an alcohol. Because the bond is more polar, there will be stronger H-bonding with the stationary phase, and the carboxylic acid will move less. This makes (D) the right answer.
Ready to get started with MCAT? Sign up for a Free MCAT practice test today!